This afternoon, Action Duchenne has since had confirmation by the Santhera team and this article that this decision has no consequences on their …
Update on CHMP negative opinion for Raxone (idebenone)Read More

September 19, 2017 by abzali123
This afternoon, Action Duchenne has since had confirmation by the Santhera team and this article that this decision has no consequences on their …
Update on CHMP negative opinion for Raxone (idebenone)Read More

September 18, 2017 by abzali123
Phrixus Pharmaceuticals has announced that the first person living with Duchenne has completed 15 months of treatment with Carmeseal-MD. The …
First person reaches 15 month mark through Phrixus’ Carmseal-MDRead More
September 12, 2017 by abzali123
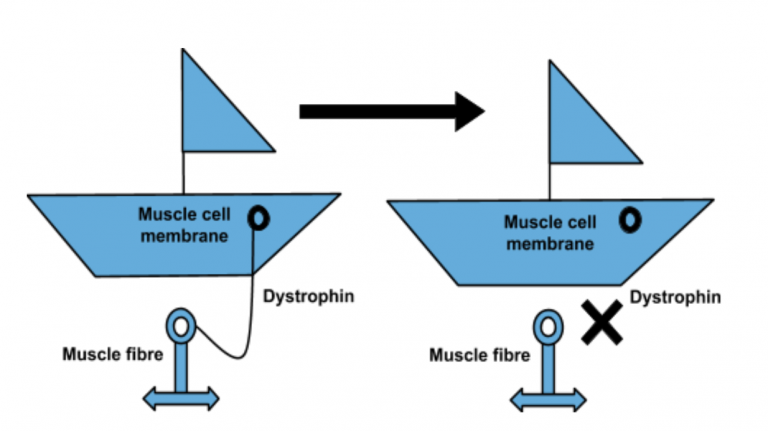
Promising microdystrophin pre-clinical work, moving towards a potential therapy has shown to restore muscle function in the canine model (dogs). …

September 12, 2017 by abzali123
Parallel London 2017 was an absolutely inspirational day! 60 incredible people took part for Action Duchenne, representing the entire community, …

September 12, 2017 by abzali123
Biophytis clinical stage drug-candidate Sarconeos demonstrates efficacy in preclinical models of Duchenne muscular dystrophy An abstract …
Sarconeos announced as new potential treatment for DuchenneRead More

September 11, 2017 by abzali123
We are delighted to announce the first people are enrolled in the UK's Early Access to Medicines Scheme (EAMS) for Raxone. Janet Bloor, Chair of …
Breaking news – first people enrolled in Raxone EAMS scheme in UKRead More

September 7, 2017 by abzali123
Kathy Wedell, mother to Isaac, is Action Duchenne's new Campaigns Officer "On World Duchenne Awareness Day I’m delighted to announce that …
Action Duchenne are Delighted to Announce Our New Campaigns Officer: KathyRead More

August 24, 2017 by abzali123
On Friday 10 November 2017, the first day of our new and extended international conference will be dedicated to new horizons in pre-clinical research …

August 14, 2017 by abzali123
NICE (the National Institute for Health and Care Excellence) have released for consultation, a new set of draft guidelines for healthcare …
NICE Consultation on Guidelines for Suspected Neurological ConditionsRead More
August 14, 2017 by abzali123
A number of pharmaceutical and biotech companies working in the field of Duchenne muscular dystrophy, have recently reported their 2nd quarter …

August 14, 2017 by abzali123
In the seventh interview in the “Portrait of Duchenne” series, our Canadian partners, La Force talked with Ravi Mehta at the Action …

August 14, 2017 by abzali123
Researchers at Cardiff University, in collaboration with biotechnology company PerkinElmer, have created a more reliable test to screen newborn babies …
Test with the Potential to More Reliably Screen for Duchenne in Newborns is DevelopedRead More
Action Duchenne
5th Floor, Mariner House
62 Prince Street
Bristol
BS1 4QD
07535 498 506
info@actionduchenne.org
